Quantum numbers5/27/2023  
These wave functions are referred to as hydrogen-like wave functions because they apply to any one-electron system (e.g., He +, Li 2+, etc.). The solutions are described in detail in most quantum mechanics books so it is not necessary to solve the equations here (see suggested readings at the end of this chapter), but Table 2.2 shows the wave functions. Even after this is done, the resulting equations are quite complex and the solution of two of the three equations requires the use of a series techniques to solve them. When the transformation is made, the variables can be separated so that three second-order differential equations are obtained with each containing one coordinate as the variable. After that is done, the Laplacian must be transformed into polar coordinates, which is a tedious task. To circumvent this problem, a change of coordinates to polar coordinates be made. However, with r expressed as the square root of the sum of the squares of the three variables it is impossible to separate the variables. The usual technique for solving such an equation is to use a procedure known as the separation of variables. The wave equation is a second-order partial differential equation in three variables. Physics was progressing at a rapid pace at that time. His adaptation consisted of representing the wave motion of an electron by means of the de Broglie relationship that had been established only two years earlier. Schrödinger did not derive a wave equation. In this model, a sphere was assumed to be covered with water, and the problem was to deal with the wave motion that would result if the surface were disturbed. His starting point was a three-dimensional wave equation that had been developed earlier by physicists who were dealing with the so-called flooded planet problem. Because of the nature of this book, we will progress directly to the problem of the hydrogen atom that was solved in 1926 by Erwin Schrödinger. All these models provide additional insight to the methods of quantum mechanics, and the interested reader should consult a quantum mechanics text such as those listed in the references at the end of this chapter. These include the particle in a one-dimensional box, the particle in a three-dimensional box, the rigid rotor, the harmonic oscillator, and barrier penetration. 
There are several elementary problems that can be solved exactly by quantum mechanical methods. 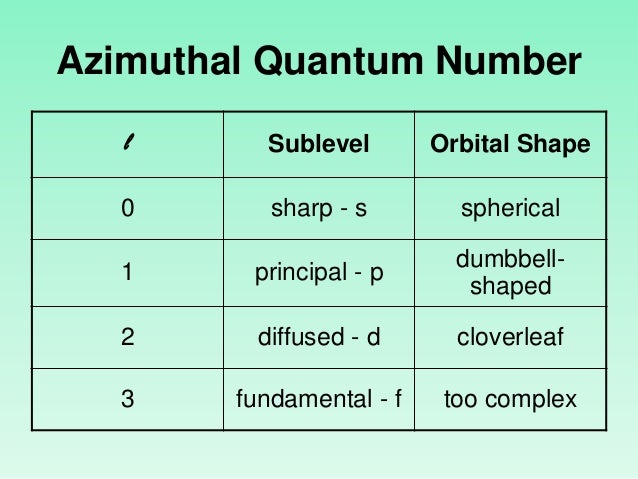
House, in Inorganic Chemistry (Third Edition), 2020 2.2 The hydrogen atom 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
